Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Yotsuji, Kenji*; Tachi, Yukio; Sakuma, Hiroshi*; Kawamura, Katsuyuki*
Applied Clay Science, 204, p.106034_1 - 106034_13, 2021/04
Times Cited Count:65 Percentile:99.67(Chemistry, Physical) on Na-montmorillonite studied by time-resolved laser fluorescence spectroscopy and surface complexation modeling
on Na-montmorillonite studied by time-resolved laser fluorescence spectroscopy and surface complexation modelingSasaki, Takayuki*; Ueda, Kenyo*; Saito, Takumi; Aoyagi, Noboru; Kobayashi, Taishi*; Takagi, Ikuji*; Kimura, Takaumi; Tachi, Yukio
Journal of Nuclear Science and Technology, 53(4), p.592 - 601, 2016/04
Times Cited Count:12 Percentile:74.37(Nuclear Science & Technology)The influences of pH and the concentrations of Eu and NaNO
and NaNO on the sorption of Eu
on the sorption of Eu to Na-montmorillonite were investigated through batch sorption measurements and time-resolved laser fluorescence spectroscopy (TRLFS). The pH had a little effect on the distribution coefficients (Kd) in 0.01 M NaNO
to Na-montmorillonite were investigated through batch sorption measurements and time-resolved laser fluorescence spectroscopy (TRLFS). The pH had a little effect on the distribution coefficients (Kd) in 0.01 M NaNO , whereas the Kd strongly depended on pH at 1 M NaNO
, whereas the Kd strongly depended on pH at 1 M NaNO . A cation exchange model combined with a one-site non-electrostatic surface complexation model was successfully applied to the measured Kd. The TRLFS spectra of Eu
. A cation exchange model combined with a one-site non-electrostatic surface complexation model was successfully applied to the measured Kd. The TRLFS spectra of Eu sorbed were processed by parallel factor analysis (PARAFAC), which corresponded to one outer-sphere (factor A) and two inner-sphere (factor B and C) complexes. It turned out that factors A and B correspond to Eu
sorbed were processed by parallel factor analysis (PARAFAC), which corresponded to one outer-sphere (factor A) and two inner-sphere (factor B and C) complexes. It turned out that factors A and B correspond to Eu sorbed by ion exchange sites and inner-sphere complexation with hydroxyl groups of the edge faces, respectively. Factor C became dominant at relatively high pH and ionic strength and likely correspond to the precipitation of Eu(OH)
sorbed by ion exchange sites and inner-sphere complexation with hydroxyl groups of the edge faces, respectively. Factor C became dominant at relatively high pH and ionic strength and likely correspond to the precipitation of Eu(OH) on the surface.
on the surface.
Zhao, Y.; Yoshimura, Kimio; Shishitani, Hideyuki*; Yamaguchi, Susumu*; Tanaka, Hirohisa*; Koizumi, Satoshi*; Szekely, N.*; Radulescu, A.*; Richter, D.*; Maekawa, Yasunari
Soft Matter, 12(5), p.1567 - 1578, 2016/02
Times Cited Count:27 Percentile:80.24(Chemistry, Physical) from synthetic, pharmaceutical and alloy samples
from synthetic, pharmaceutical and alloy samplesNaushad, M.*; Alothman, Z. A.*; Alam, M. M.*; Awual, M. R.; Eldesoky, G. E.*; Islam, M.*
Journal of the Iranian Chemical Society, 12(9), p.1677 - 1686, 2015/09
Times Cited Count:61 Percentile:83.92(Chemistry, Multidisciplinary) resolution
resolutionChatake, Toshiyuki*; Kurihara, Kazuo; Tanaka, Ichiro*; Tsyba, I.*; Bau, R.*; Jenney, F. E. Jr.*; Adams, M. W. W.*; Niimura, Nobuo
Acta Crystallographica Section D, 60(8), p.1364 - 1373, 2004/08
Times Cited Count:34 Percentile:88.87(Biochemical Research Methods)A neutron diffraction study has been carried out at 1.6  resolution on a mutant rubredoxin from
resolution on a mutant rubredoxin from 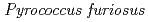 using the BIX-3 single-crystal diffractometer at the JRR-3 reactor of JAERI. In order to study the unusual thermostability of rubredoxin from
using the BIX-3 single-crystal diffractometer at the JRR-3 reactor of JAERI. In order to study the unusual thermostability of rubredoxin from 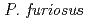 , the hydrogen-bonding patterns were compared between the native and a 'triple-mutant' variant where three residues were changed so that they are identical to those in a mesophilic rubredoxin. In the present study, some minor changes were found between the wild-type and mutant proteins in the hydrogen-bonding patterns of the Trp3/Tyr3 region. The H/D-exchange ratios in the protein were also studied. The results suggest that the backbone amide bonds near the four Cys residues of the FeS
, the hydrogen-bonding patterns were compared between the native and a 'triple-mutant' variant where three residues were changed so that they are identical to those in a mesophilic rubredoxin. In the present study, some minor changes were found between the wild-type and mutant proteins in the hydrogen-bonding patterns of the Trp3/Tyr3 region. The H/D-exchange ratios in the protein were also studied. The results suggest that the backbone amide bonds near the four Cys residues of the FeS redox center are most resistant to H/D exchange. In addition, the 1.6
redox center are most resistant to H/D exchange. In addition, the 1.6  resolution of the present neutron structure determination has revealed a more detailed picture than previously available of some portions of the water structure, including ordered and disordered O-D bonds.
resolution of the present neutron structure determination has revealed a more detailed picture than previously available of some portions of the water structure, including ordered and disordered O-D bonds.
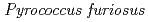 by BIX-3, a single-crystal diffractometer for biomacromolecules
by BIX-3, a single-crystal diffractometer for biomacromoleculesKurihara, Kazuo; Tanaka, Ichiro*; Chatake, Toshiyuki*; Adams, M. W. W.*; Jenney, F. E. Jr.*; Moiseeva, N.*; Bau, R.*; Niimura, Nobuo
Proceedings of the National Academy of Sciences of the United States of America, 101(31), p.11215 - 11220, 2004/08
Times Cited Count:48 Percentile:61.07(Multidisciplinary Sciences)The structure of a rubredoxin (Rd) from 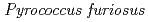 , an organism that grows optimally at 100
, an organism that grows optimally at 100  C, was determined using the neutron single-crystal diffractometer for biological macromolecules (BIX-3) at the JRR-3 reactor of JAERI. Data were collected at room temperature up to a resolution of 1.5
C, was determined using the neutron single-crystal diffractometer for biological macromolecules (BIX-3) at the JRR-3 reactor of JAERI. Data were collected at room temperature up to a resolution of 1.5  , and the completeness of the data set was 81.9 %. The model contains 306 H atoms and 50 D atoms. A total of 37 hydration water molecules were identified. The model has been refined to final agreement factors of
, and the completeness of the data set was 81.9 %. The model contains 306 H atoms and 50 D atoms. A total of 37 hydration water molecules were identified. The model has been refined to final agreement factors of 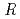 = 18.6 % and
= 18.6 % and 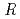
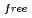 = 21.7 %. Several orientations of the O-D bonds of side chains, whose assignments from X-ray data were previously ambiguous, were clearly visible in the neutron structure. While most backbone N-H bonds had undergone some degree of H/D exchange throughout the molecule, five H atom positions still had distinctly negative (H) peaks. The neutron Fourier maps clearly showed the details of an extensive set of H bonds involving the ND
= 21.7 %. Several orientations of the O-D bonds of side chains, whose assignments from X-ray data were previously ambiguous, were clearly visible in the neutron structure. While most backbone N-H bonds had undergone some degree of H/D exchange throughout the molecule, five H atom positions still had distinctly negative (H) peaks. The neutron Fourier maps clearly showed the details of an extensive set of H bonds involving the ND
 terminus that may contribute to the unusual thermostability of this molecule.
terminus that may contribute to the unusual thermostability of this molecule.
 FeCoO
FeCoO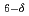 and SrBaFeCoO
and SrBaFeCoO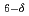 ; Effects induced by magnetic order and transport coherence
; Effects induced by magnetic order and transport coherenceRykov, A. I.*; Nomura, Kiyoshi*; Sawada, Tsuguo*; Mitsui, Takaya; Seto, Makoto*; Tamegai, Tsuyoshi*; Tokunaga, Masashi*
Physical Review B, 68(22), p.224401_1 - 224401_7, 2003/12
Times Cited Count:8 Percentile:42.85(Materials Science, Multidisciplinary)no abstracts in English
Arifal; Hwang, G.; Onuki, Kaoru
Journal of Membrane Science, 210(1), p.39 - 44, 2002/12
Times Cited Count:19 Percentile:59.78(Engineering, Chemical)no abstracts in English
Yamagishi, Isao
JAERI-Review 2001-027, 52 Pages, 2001/07
The present study deals with the survey of inorganic ion-exchangers suitable for separation and immobilization of Sr and Cs contained in acidic high-level liquid waste. For separation and immobilization of Cs, crystalline silicotitanate seems to be the most promising exchanger. For selective separation of Sr, there is no promising exchanger up to now. Sintered ceramics are favorable waste forms of inorganic ion-exchangers. Their stabilities are largely influenced by composition of exchangers, qualities of ceramics and disposal scenario.
*; Hirata, Masaru; Yahata, Taneaki
Journal of Nuclear Science and Technology, 28(3), p.228 - 238, 1991/03
no abstracts in English
 Sr using liquid scintillation counting techniques
Sr using liquid scintillation counting techniquesAmano, Hikaru
Proc. of the 4th Int. Seminor of Liquid Scintillation Analysis, p.236 - 245, 1991/00
no abstracts in English
Yahata, Taneaki; *; Hirata, Masaru
JAERI-M 90-075, 68 Pages, 1990/05
no abstracts in English
 -HI mixed acid solution
-HI mixed acid solutionUsuda, Shigekazu; Sakurai, Satoshi; Hirata, Masaru; Umezawa, Hirokazu
Sep. Sci. Technol., 25(11-12), p.1225 - 1237, 1990/00
Times Cited Count:1 Percentile:27.36(Chemistry, Multidisciplinary)no abstracts in English
; ; *
Journal of Radioanalytical and Nuclear Chemistry, 109(2), p.353 - 361, 1987/02
Times Cited Count:7 Percentile:60.01(Chemistry, Analytical)no abstracts in English
; ; *; ;
Journal of Radioanalytical and Nuclear Chemistry, 116(1), p.125 - 132, 1987/01
Times Cited Count:3 Percentile:38.12(Chemistry, Analytical)no abstracts in English
Inorg.Chim.Acta, 140, p.279 - 282, 1987/00
no abstracts in English
Journal of Nuclear Science and Technology, 22(5), p.398 - 405, 1985/00
Times Cited Count:6 Percentile:64.59(Nuclear Science & Technology)no abstracts in English
Ozawa, K.; *; Kawatsura, Kiyoshi; ; *; *; *; *
Nuclear Instruments and Methods in Physics Research B, 9, p.621 - 625, 1985/00
Times Cited Count:3 Percentile:53.91(Instruments & Instrumentation)no abstracts in English
; Saito, Keiichiro; Shiba, Koreyuki
Journal of Nuclear Science and Technology, 20(5), p.439 - 440, 1983/00
Times Cited Count:53 Percentile:99.42(Nuclear Science & Technology)no abstracts in English
; ; *
Nucl.Chem.Waste Manage., 3, p.131 - 137, 1982/00
no abstracts in English